In a case it described as “‘a prime example’ of when ODP does not apply,” the Federal Circuit recently reversed a decision from the District of Delaware that invalidated a claim for obviousness-type double patenting (ODP), holding that a first-filed, first-issued, later-expiring claim cannot be invalidated for ODP based on a later-filed, later-issued, but earlier-expiring claim from the same family. In so doing, the court answered the question of whether a later-filed, later-issued patent in the same family can be an ODP reference against the first application in the family—it cannot. The court also clarified the scope of its prior ruling in In re Cellect, explaining that case answered a different question—the question of what expiration date should be used for an ODP analysis.
In this case, Allergan sued Sun Pharmaceuticals under the Hatch-Waxman Act after Sun submitted an Abbreviated New Drug Application (ANDA) to market a generic version of the drug Viberzi. One of the asserted patents, U.S. Patent No. 7,741,356 (“the ’356 patent”), had been granted 4671 days of patent term adjustment (PTA) for delays during its prosecution. As a result and as shown in the diagram below, the ’356 patent had an expiration date that was 467 days later than two later-filed and later-issued patents in the same family.
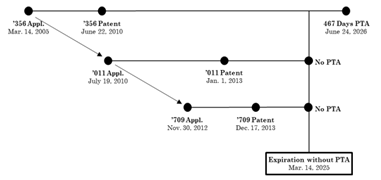
In the district court, Sun challenged the validity of claim 40 of the ’356 patent on ODP grounds, relying on the later-filed, later-issued patents as reference patents. Specifically, Sun argued that because the claims of the three patents are not patentably distinct and because claim 40 of the ’356 patent has a later expiration date, it must be invalid for ODP. The district court agreed with Sun and found claim 40 invalid based solely on its later expiration date.
On appeal, Allergan argued that the district court misunderstood Cellect, contending that this case presented a different scenario. According to Allergan, because the ’356 patent was the first patent to be filed and first patent to issue in the family, it is not subject to ODP challenges over the later-filed, later-issued patents. And consequently, the rule of Cellect simply does not apply.
The Federal Circuit agreed with Allergan, explaining that Cellect established a rule whereby PTA is included in the expiration date when conducting an ODP analysis. Cellect did not, however, define the circumstances under which a patent can properly serve as an ODP reference. In particular, Cellect did not consider whether a first-filed, first-issued patent that receives PTA can be invalidated for ODP based on a later-filed patent with the same priority date. As to that question, the Federal Circuit held in this case that it cannot. To rule otherwise, would be “antithetical to the principles of ODP,” which serves to prevent a patentee from obtaining a second later-expiring patent directed to patentably indistinct subject matter. Thus, where the later-expiring patent is the first patent in the family to be filed and the first patent in the family to issue, PTA does not extend the period of exclusivity—it sets the term.
Allergan USA, Inc. v. MSN Laboratories Private Ltd., C.A. No. 2024-1061 (August 13, 2024)
1The USPTO initially awarded 1,107 days of PTA to the ’356 patent, but all but 467 days were disclaimed as part of an application for patent term extension (PTE).


